
Proteome-wide discovery at the scale your research demands
Protein measurements across thousands of samples. Absolute quantification. Pricing built for discovery.
The Nomic Platform makes high-throughput profiling possible for drug discovery
Cost Efficient
Generate large proteomic datasets cost-effectively (~$50/sample), enabling well-powered studies.
Quantitative & Interoperable
Generate standardized data (pg/mL) that seamlessly integrate across studies and into workflows.
Proteome-Wide & Scalable
Measure over 1000 proteins expertly selected to capture biologically meaningful signals at scale.
Proteomics driving
discovery
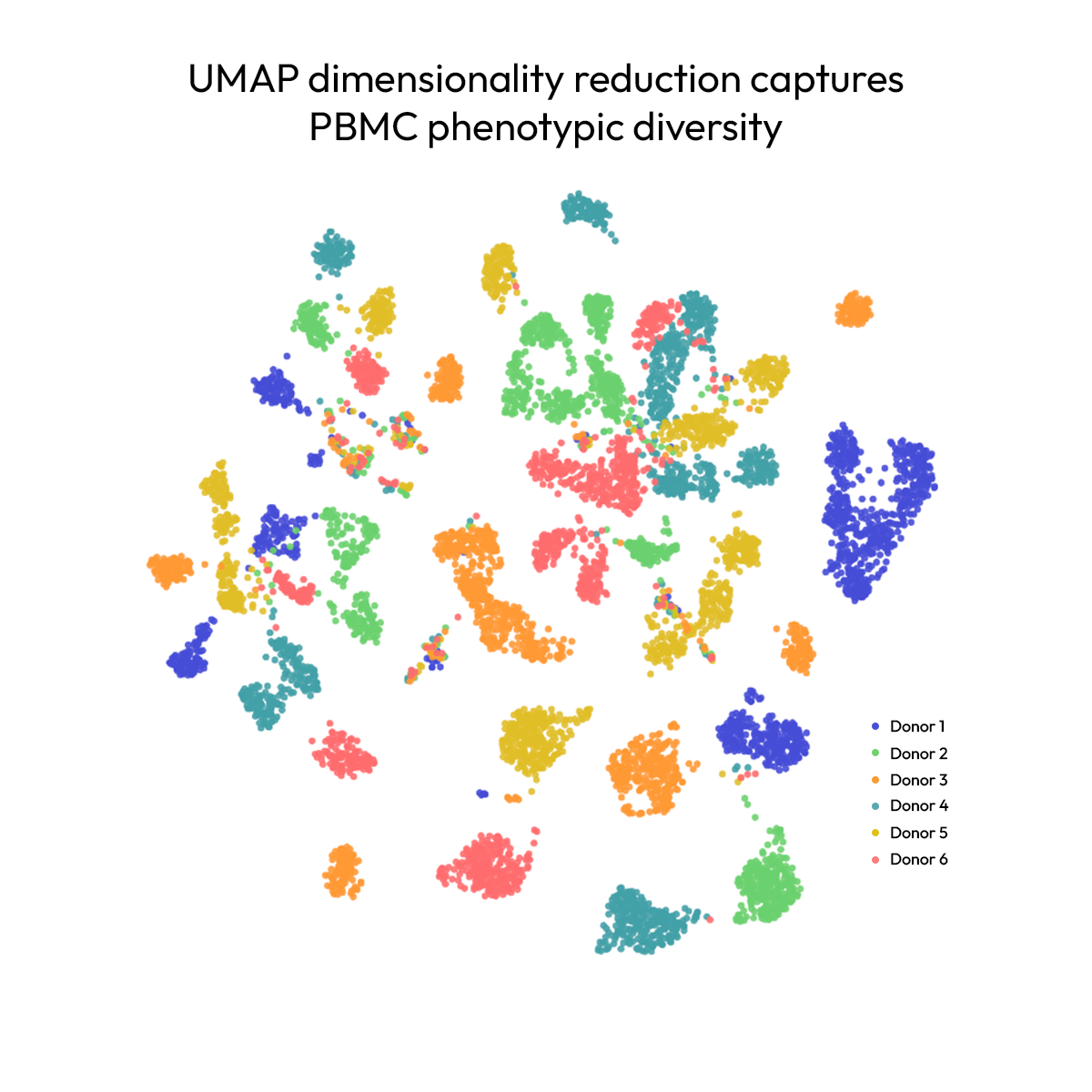
Capturing the cytokine biology that drives better drug decisions
A comprehensive map of cytokine signaling has long been missing from drug discovery — transcriptomics provides a valuable proxy, but cytokine biology is largely regulated at the protein level, leaving critical gaps in what gene expression alone can reveal.
In a large-scale PBMC perturbation dataset, Nomic's high-plex secretome profiling reveals that many critical cytokine responses are not captured at the RNA level. Across thousands of samples, protein-level measurements uncover regulatory mechanisms that gene expression alone cannot capture — providing a more complete and reliable foundation for immunology drug decisions.
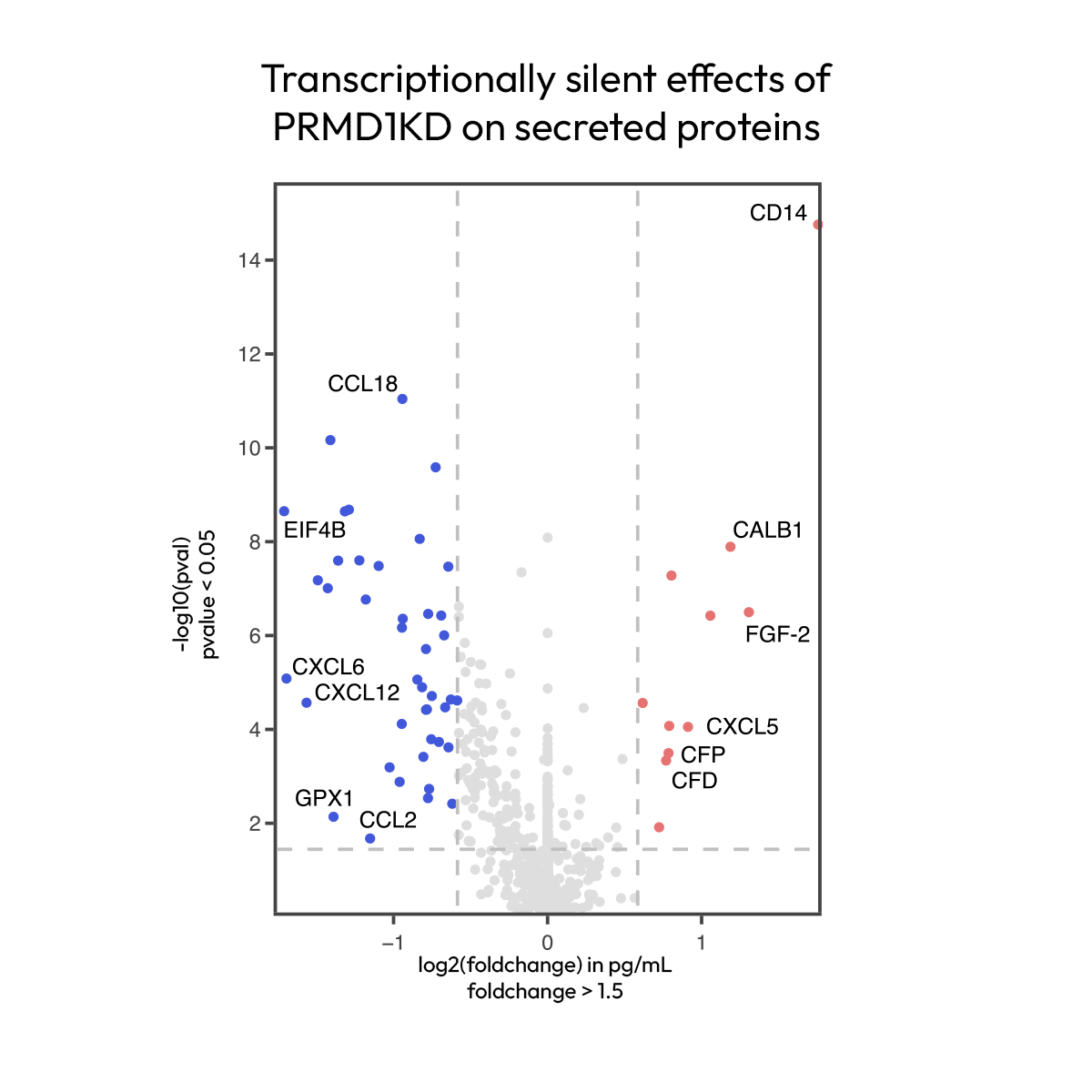
Revealing protein-only phenotypes in functional genomic screens
Understanding how genes regulate cell phenotypes requires scalable measurement of functional outputs; while some activities are well represented by mRNA proxies, others require more direct protein readouts. In CRISPRi screens of iPSC-derived microglia, Nomic Omni 1000 reveals that chemokines are almost exclusively regulated at the protein level, as are markers of cell death and receptor shedding. This provides a direct readout of immune function that transcriptomic approaches fail to capture.
Data generated in collaboration with The Martin Kampmann lab at University of California, San Francisco
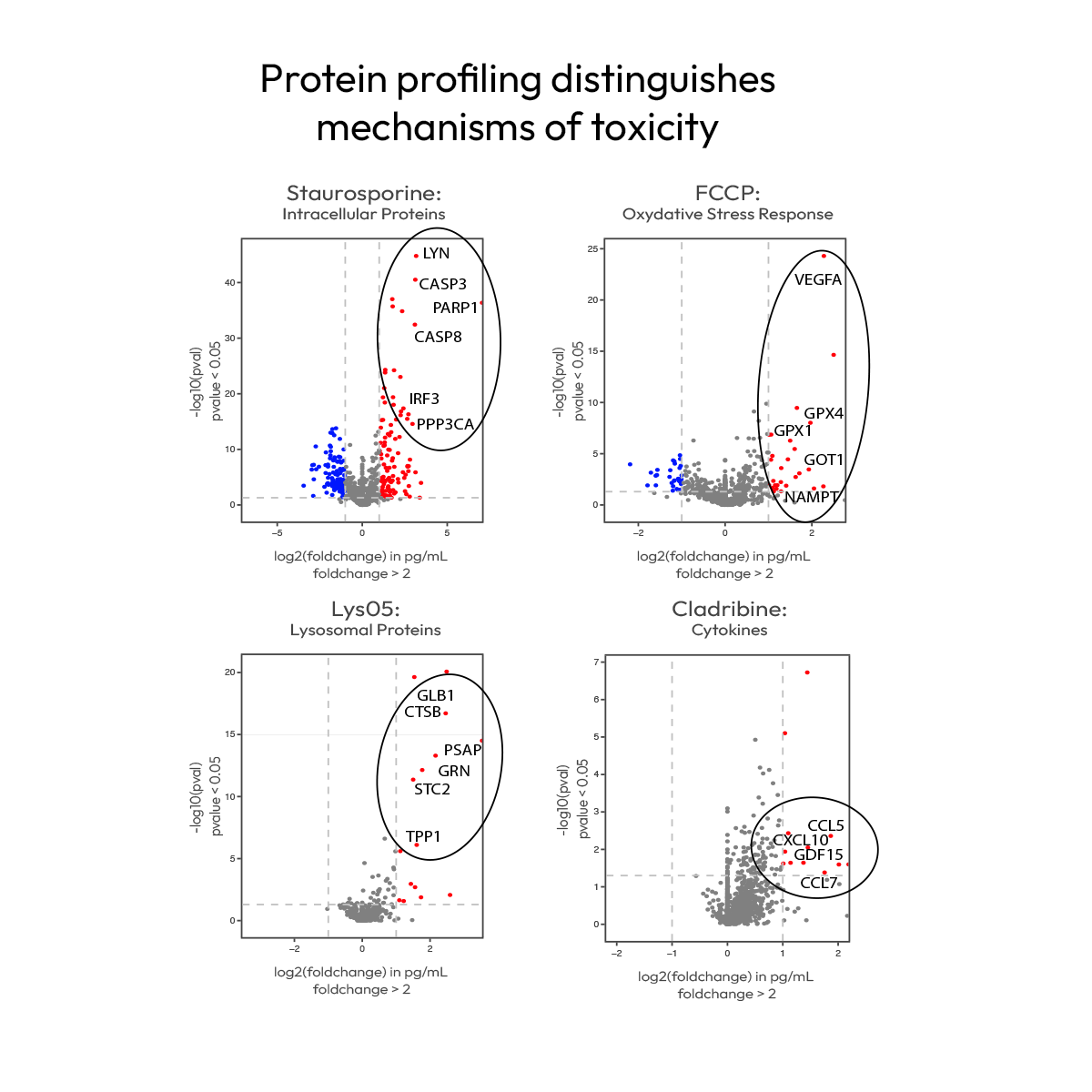
Earlier detection and deeper insights for toxicity assesment
Sensitive detection of toxic effects requires capturing both early signals and underlying biology. In large-scale toxicity studies, Nomic's Omni 1000 provides better sensitivity than conventional assays and goes beyond detection to provide insight into the mechanisms driving the observed toxicity.
Protein signatures captured by Omni 1000 resolve distinct mechanisms of toxicity, including immunotoxic, cytotoxic, mitochondrial, and lysosomal effects. By distinguishing these responses with a single, sensitive assay, it enables a more precise understanding of compound impact and more confident safety decisions.
Proteomics that supports you all the way
From discovery screening to drug R&D and clinical studies, Nomic provides a service that scales with your projects. Whether using Omni 1000, context-specific Core options, or even a tailored Flex panel designed to your objectives, you can adapt your approach without compromising data quality or cross study compatibility.
Discovery
- Target ID
- Therapeutic Biomarker Discovery
- Indication Selection
- Target Validation
- Perturbation Biology
- MoA Studies
- Early Tox Profiles
Drug R&D
- Target Engagement
- Hit-to-Lead
- Hit Optimization
- ADME/Tox
- Pre-clinical/IND Enabling
Translational
- Biomarket Validation
- Patient Stratification
- PK/PD
- Safety
- Disease Prognosis
Stop inferring biology. Start measuring it.
See how quantitative protein data drives decisions from target selection to patient stratification.


Resources for discovery teams
Your research project deserves a protein answer
Our scientists have designed proteomics studies across target ID, perturbation biology, MoA, and early toxicity. Share the specifics of your program, and we will tell you what is possible.

